IN PATIENTS WITH ROS1+ NSCLC Up to 50% will develop resistance mutations from treatment with existing ROS1 TKIs1-3
What if you could transform your treatment approach?
Scroll down


RESISTANCE DRIVES PROGRESSION
Today’s first-line ROS1 TKIs lack the broad clinical benefit and durability to keep ROS1+ NSCLC from progressing1,4
~33% of patients don’t respond to first-line treatment with existing ROS1 TKIs5,6
Up to 50% of patients develop resistance mutations from treatment with existing ROS1 TKIs1-3
36%
~19-36% of patients present with brain metastases at baseline7,8
Getting ahead of disease progression remains a challenge
ROS1 RESISTANCE MUTATIONS
Acquired ROS1 resistance mutations drive treatment failure and disease progression1,4
- Up to 1 in 2 patients will develop resistance mutations from treatment with existing ROS1 TKIs1-3
- 1 in 3 patients will develop G2032R, the most commonly acquired ROS1 resistance mutation, after disease progression3,4,9*
*After treatment with crizotinib, entrectinib, or lorlatinib.
The 2 primary types of resistance mutations2,4,10-13
Solvent front mutations
Form at the front of the binding pocket
Gatekeeper mutations
Form at the back of the binding pocket
BINDING POCKET
ROS1 kinase domain
For illustrative purposes only.
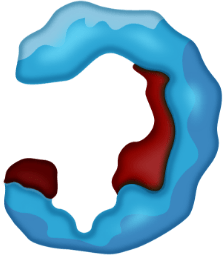
Today’s first-line ROS1 TKIs were not designed to address resistance mutations3,10,14,15
ROS1 TKI DESIGN LIMITATIONS
Approved TKI treatments do not address both brain metastases and the most common ROS1 resistance mutations1,4,7
- Treatment with existing first-line ROS1 TKIs leads to resistance mutations1-4,16
- Acquired ROS1 resistance mutations develop in the binding pocket1,2,4,10
- Current ROS1 TKIs are unable to bind effectively in the presence of resistance mutations10,17
CURRENT TKIs
For illustrative purposes only.
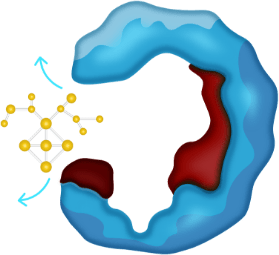
36%
- The brain is a primary site of metastasis7,18
- ~19-36% of patients present with brain metastases at baseline7,8
- Not all approved ROS1 TKIs demonstrate CNS activity5,8,19
Could a first-line TKI that’s designed differently change outcomes for patients with ROS1+ NSCLC?
THE NEXT GENERATION
Research is investigating the potential that the Next-Generation TKIs may have on progression of the disease20
- Smaller, more compact TKIs that fit precisely in their binding pockets may be able to address the most commonly acquired resistance mutations3,4,10,20,21
NEXT-GENERATION TKIs
For illustrative purposes only.
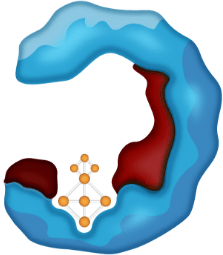
BMS is committed to researching areas of high unmet medical needs, including potential disease progression from resistance mutations

References: 1. Almquist D, Ernani V. The road less traveled: a guide to metastatic ROS1-rearranged non-small-cell lung cancer. JCO Oncol Pract. 2021;17(1):7-14. doi:10.1200/OP.20.00819 2. Gainor JF, Tseng D, Yoda S, et al. Patterns of metastatic spread and mechanisms of resistance to crizotinib in ROS1-positive non-small-cell lung cancer. JCO Precis Oncol. 2017;2017:PO.17.00063. doi:10.1200/PO.17.00063 3. Lin JJ, Choudhury NJ, Yoda S, et al. Spectrum of mechanisms of resistance to crizotinib and lorlatinib in ROS1 fusion-positive lung cancer. Clin Cancer Res. 2021;27(10):2899-2909. doi:10.1158/1078-0432.CCR-21-0032 4. D’Angelo A, Sobhani N, Chapman R, et al. Focus on ROS1-positive non-small cell lung cancer (NSCLC): crizotinib, resistance mechanisms and the newer generation of targeted therapies. Cancers (Basel). 2020;12(11):3293. doi:10.3390/cancers12113293 5. Dziadziuszko R, Krebs MG, De Braud F, et al. Updated integrated analysis of the efficacy and safety of entrectinib in locally advanced or metastatic ROS1 fusion-positive non-small-cell lung cancer. J Clin Oncol. 2021;39(11):1253-1263. doi:10.1200/JCO.20.03025 6. Stenger M. Crizotinib in ROS1-positive metastatic non-small cell lung cancer. April 10, 2016. Accessed March 30, 2022. https://ascopost.com/issues/april-10-2016/crizotinib-in-ros1-positive-metastatic-non-small-cell-lung-cancer 7. Tan AC, Itchins M, Khasraw M. Brain Metastases in Lung Cancers with Emerging Targetable Fusion Drivers. International Journal of Molecular Sciences. 2020; 21(4):1416. https://doi.org/10.3390/ijms21041416 8. Zhang Y, Zhang X, Zhang R, et al. Clinical and molecular factors that impact the efficacy of first-line crizotinib in ROS1-rearranged non-small-cell lung cancer: a large multicenter retrospective study. BMC Med. 2021;19(1):206. doi:10.1186/s12916-021-02082-6 9. Doebele RC, Dziadziuszko R, Drilon A, et al. Genomic landscape of entrectinib resistance from ctDNA analysis in STARTRK-2. Abstract presented at: 44th ESMO Congress; September 27 – October 1, 2019; Barcelona, Spain. Late-breaking and deferred publication abstracts. 10. Roskoski R Jr. ROS1 protein-tyrosine kinase inhibitors in the treatment of ROS1 fusion protein-driven non-small cell lung cancers. Pharmacol Res. 2017;121:202-212. doi:10.1016/j.phrs.2017.04.022 11. Cui JJ. A new challenging and promising era of tyrosine kinase inhibitors. ACS Med Chem Lett. 2014;5(4):272-274. doi:10.1021/ml500091p 12. Mondal J, Tiwary P, Berne B-J. How a kinase inhibitor withstands gatekeeper residue mutations. J Am Chem Soc. 2016;138(13):4608-4615. doi:10.1021/jacs.6b01232 13. Lin JJ, Shaw AT. Recent advances in targeting ROS1 in lung cancer. J Thorac Oncol. 2017;12(11):1611-1625. doi:10.1016/j.jtho.2017.08.002 14. Ardini E, Menichincheri M, Banfi P, et al. Entrectinib, a pan-TRK, ROS1, and ALK inhibitor with activity in multiple molecularly defined cancer indications. Mol Cancer Ther. 2016;15(4):628-639. doi:10.1158/1535-7163.MCT-15-0758 15. Menichincheri M, Ardini E, Magnaghi P, et al. Discovery of entrectinib: a new 3-aminoindazole as a potent anaplastic lymphoma kinase (ALK), c-ros oncogene 1 kinase (ROS1), and pan-tropomyosin receptor kinases (pan-TRKs) inhibitor. J Med Chem. 2016;59(7):3392-3408. doi:10.1021/acs.jmedchem.6b00064 16. Frampton JE. Entrectinib: a review in NTRK+ solid tumours and ROS1+ NSCLC. Drugs. 2021;81(6):697-708. doi:10.1007/s40265-021-01503-3 17. Liu D, Offin M, Harnicar S, Li BT, Drilon A. Entrectinib: an orally available, selective tyrosine kinase inhibitor for the treatment of NTRK, ROS1, and ALK fusion-positive solid tumors. Ther Clin Risk Manag. 2018;14:1247-1252. doi:10.2147/TCRM.S147381 18. Patil T, Smith DE, Bunn PA, et al. The incidence of brain metastases in stage IV ROS1-rearranged non-small cell lung cancer and rate of central nervous system progression on crizotinib. J Thorac Oncol. 2018;13(11):1717-1726. doi:10.1016/j.jtho.2018.07.001 19. Sehgal K, Patell R, Rangachari D, Costa DB. Targeting ROS1 rearrangements in non-small cell lung cancer with crizotinib and other kinase inhibitors. Transl Cancer Res. 2018;7(suppl 7):S779-S789. doi:10.21037/tcr.2018.08.11 20. Ahnert JR, Gray N, Mok T, Gainor J. What it takes to improve a first-generation inhibitor to a second- or third-generation small molecule. Am Soc Clin Oncol Educ Book. 2019;39:196-205. doi:10.1200/EDBK_242209 21. Abourehab M A-S, Alqahtani AM, Youssif B G-M, Gouda AM. Globally approved EGFR inhibitors: insights into their syntheses, target kinases, biological activities, receptor interactions, and metabolism. Molecules. 2021;26(21):6677. doi:10.3390/molecules26216677